-
Relacorilant Plus Nab-Paclitaxel Extends Survival in Women with Recurrent, Platinum-resistant Ovarian Cancer
Source: Nasdaq GlobeNewswire / 30 Mar 2022 13:05:00 America/Los_Angeles
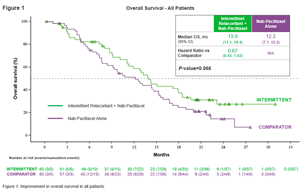
- Addition of relacorilant to nab-paclitaxel reduces risk of death by 33 percent compared to treatment with nab-paclitaxel alone (p-value: 0.066), without increased side effects
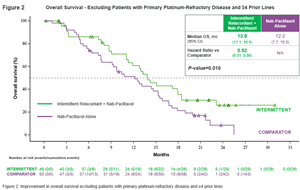
- Excluding patients (i) who had not responded to even their first treatment with a platinum-based therapy (“primary platinum-refractory” patients) or (ii) who had received four or more prior lines of therapy, addition of relacorilant to nab-paclitaxel significantly reduces risk of death by 48 percent compared to treatment with nab-paclitaxel alone (p-value: 0.010)
- Phase 3 trial, which will exclude patients with primary platinum-refractory disease and who have had four or more prior lines of therapy, is planned to open in second quarter of 2022
MENLO PARK, Calif., March 30, 2022 (GLOBE NEWSWIRE) -- Corcept Therapeutics Incorporated (NASDAQ: CORT), a commercial stage company engaged in the discovery, development and sale of drugs to treat severe metabolic, oncologic and neuropsychiatric disorders by modulating the effects of the hormone cortisol, today announced overall survival (“OS”) data from its 178-patient, randomized, controlled, Phase 2 study of relacorilant plus nab-paclitaxel in patients with recurrent, platinum-resistant ovarian cancer.
Women treated with relacorilant the day before, the day of and the day after their regular nab-paclitaxel infusions (the trial’s “intermittent” arm) experienced a 33% reduction in risk of death compared to women treated with nab-paclitaxel alone (hazard ratio: 0.67; p-value: 0.066). Their median OS was 13.9 months, compared to 12.2 months for women receiving nab-paclitaxel monotherapy. (See Figure 1)
The women who enrolled in this trial were very sick and had experienced disease progression following prior therapy (median number of prior therapies: three). By chance, more women with either primary platinum-refractory disease or who had already received four or more prior lines of therapy, both indicators of a very poor prognosis, were assigned to the intermittent arm. As is typical of late-stage clinical trials, such women will not be enrolled in Corcept’s upcoming Phase 3 trial.
Excluding primary platinum-refractory patients and women who had already received four or more prior lines of therapy, women treated with relacorilant intermittently experienced a 48% reduced risk of death compared to women treated with nab-paclitaxel alone (hazard ratio: 0.52; p-value: 0.010). Their median OS was 13.9 months, compared to 12.2 months for women receiving nab-paclitaxel monotherapy. (See Figure 2)
“Corcept has introduced a novel oncologic therapeutic platform, cortisol modulation. These results constitute a potentially important medical advance,” said Thomas Herzog, MD, Deputy Director at the University of Cincinnati Cancer Center, member of the Board of Directors of the Gynecologic Oncology Group (GOG) Foundation and Associate Director of GOG Partners. “In this large, randomized study, women with recurrent, platinum-resistant ovarian cancer who were administered relacorilant at the time they received nab-paclitaxel exhibited meaningful improvements in progression free survival, duration of response and overall survival – without increased side effects – compared to women who received nab-paclitaxel alone. For this patient population, relacorilant plus nab-paclitaxel has the potential to become a new standard of care.”
“We are excited to receive these survival data, which have continued to improve as the trial has progressed,” said Bill Guyer, PharmD, Corcept’s Chief Development Officer. “If our Phase 3 trial replicates the results in progression free survival, duration of response and overall survival that we’ve seen in Phase 2, it will be an unprecedented success for patients with ovarian cancer. No approved therapy has been shown to significantly extend survival compared to standard chemotherapy in women with platinum-resistant ovarian cancer. We plan to meet with the FDA as soon as possible to define the best path forward and to open our Phase 3 trial in the second quarter of 2022.”
On March 31, 2022, Corcept will host an event for investors and analysts regarding its ovarian cancer program. Click here to register. In addition, the company plans to present these results at a major oncology conference in 2022.
About Platinum-Resistant Ovarian Cancer
Ovarian cancer is the fifth most common cause of cancer death in women.1 Patients whose disease returns less than six months after receiving platinum-containing therapy are described as having “platinum-resistant” disease. In the United States, approximately 20,000 women with platinum-resistant disease are candidates to start a new therapy each year.2 There are few treatment options and median overall survival following recurrence of disease is 12 months or less with single-agent chemotherapy.3 No approved therapy has been shown to significantly extend overall survival in patients with recurrent, platinum-resistant ovarian cancer compared to standard chemotherapy.4
About Corcept’s Ovarian Cancer Program
The data in this release come from Corcept’s 178-patient, randomized, controlled, Phase 2 trial of relacorilant plus nab-paclitaxel in patients with recurrent, platinum-resistant ovarian cancer.5 The women who entered the trial had experienced disease progression on prior lines of therapy. The median number of prior treatments was three.
Study participants were randomized 1:1:1 to receive either (i) nab-paclitaxel plus 150 mg of relacorilant given the day before, the day of, and the day after each weekly nab-paclitaxel infusion (“intermittent” arm), (ii) nab-paclitaxel plus 100 mg relacorilant given daily (“continuous” arm) or (iii) nab-paclitaxel alone (“comparator” arm).
OS was assessed after a pre-determined number of patient deaths had occurred. At the time of database cutoff, 128 of the 178 women who enrolled in the study had died. Fourteen additional women in the intermittent arm and nine additional women in the comparator arm are expected to contribute to the final overall survival results. One woman in the intermittent arm and one in the continuous arm have yet to experience tumor progression – for both patients it has been over twenty months since they initiated therapy with relacorilant.
Relative to women in the comparator arm, women in the intermittent arm experienced significantly improved progression free survival (PFS) (median PFS: 5.6 months versus 3.8 months; hazard ratio: 0.66; p-value: 0.038) and duration of response (DoR) (median DoR: 5.6 months versus 3.7 months, hazard ratio: 0.36; p-value: 0.006). They also lived longer (median OS: 13.9 months versus 12.2 months, hazard ratio: 0.67; p-value: 0.066). Safety and tolerability of relacorilant plus nab-paclitaxel was comparable to nab-paclitaxel monotherapy. (See Table 1)
Phase 2 Trial Results – Intermittent and Comparator Arms (All Patients) Intermittent Comparator Intermittent vs Comparator Median PFS* (95% CI) 5.6 months 3.8 months Hazard Ratio: 0.66 (0.44, 0.98)
p-value: 0.038Median DoR* (95% CI) 5.6 months 3.7 months Hazard Ratio: 0.36 (0.16, 0.77)
p-value: 0.006Median OS* (95% CI) 13.9 months 12.2 months Hazard Ratio: 0.67 (0.43, 1.03)
p-value: 0.066Table 1: Summary of results of intermittent and comparator arms – all patients.
*PFS: progression free survival; OS: overall survival; DoR: duration of response.Excluding women with primary platinum-refractory disease and patients who had received four or more prior lines of treatment, women in the intermittent arm experienced significantly improved PFS (median PFS: 5.6 months versus 3.8 months, hazard ratio: 0.58; p-value: 0.016) and OS relative to women in the comparator arm (median OS: 13.9 months versus 12.2 months, hazard ratio: 0.52; p-value: 0.010). The women in the intermittent arm also experienced a significant improvement in DoR relative to those in the comparator arm (median DoR: 5.6 months versus 3.6 months, hazard ratio: 0.26; p-value: 0.001). Safety and tolerability of relacorilant plus nab-paclitaxel was comparable to nab-paclitaxel monotherapy. (See Table 2)
Phase 2 Trial Results – Intermittent and Comparator Arms (Planned Phase 3 Patient Population) Intermittent Comparator Intermittent vs Comparator Median PFS* (95% CI) 5.6 months 3.8 months Hazard Ratio: 0.58 (0.37, 0.91)
p-value: 0.016Median DoR* (95% CI) 5.6 months 3.6 months Hazard Ratio: 0.26 (0.11, 0.62)
p-value: 0.001Median OS* (95% CI) 13.9 months 12.2 months Hazard Ratio: 0.52 (0.31, 0.86)
p-value: 0.010Table 2: Summary of results of intermittent and comparator arms – excluding patients with primary platinum-refractory disease and patients who had received four or more prior lines of treatment.
*PFS: progression free survival; OS: overall survival; DoR: duration of response.About Corcept’s Oncology Programs
There is substantial evidence that cortisol activity at the glucocorticoid receptor (“GR”) allows certain solid tumors to resist treatment and that modulating cortisol’s activity may help anti-cancer treatments achieve their intended effect.
Many types of solid tumors express the GR and are potential targets for cortisol modulation therapy. In some cancers, cortisol inhibits cellular apoptosis – the tumor-killing effect many treatments are meant to stimulate. In other cancers, cortisol activity promotes tumor growth. Cortisol also suppresses the body’s immune response; activating – not suppressing – the immune system is beneficial in fighting certain cancers.
Corcept is conducting clinical trials of its proprietary selective cortisol modulators in combination with three different anti-cancer treatments in patients with ovarian, adrenal and prostate cancers. Corcept’s first controlled study in oncology – relacorilant plus nab-paclitaxel for the treatment of patients with ovarian cancer – has demonstrated statistically significant and clinically meaningful results.
About Relacorilant
Relacorilant is a non-steroidal, selective glucocorticoid receptor modulator that does not bind to the body's other hormone receptors. Corcept is studying relacorilant in a variety of serious disorders, including ovarian and adrenal cancer and Cushing’s syndrome. Relacorilant is proprietary to Corcept and is protected by composition of matter and method of use patents, as well as orphan drug designation in the United States for the treatment of pancreatic cancer and both the United States and the European Union for the treatment of Cushing’s syndrome.
About Corcept Therapeutics
Corcept has discovered a large portfolio of proprietary compounds that selectively modulate the effects of cortisol and owns extensive United States and foreign intellectual property covering both their composition and their use to treat a variety of serious disorders. The company is conducting clinical trials of its leading cortisol modulators as potential treatments for patients with Cushing’s syndrome, ovarian, adrenal and prostate cancer, weight gain caused by the use of antipsychotic medications and liver disease. Corcept’s drug Korlym® was the first medication approved by the U.S. Food and Drug Administration for the treatment of patients with Cushing’s syndrome.
Forward Looking Statements
Statements in this press release, other than statements of historical fact, are forward-looking statements based on our current plans and expectations that are subject to risks and uncertainties that might cause our actual results to differ materially from those such statements express or imply. These risks and uncertainties include, but are not limited to, our ability to operate our business, conduct our clinical trials and achieve our other goals during the COVID-19 pandemic; risks related to the development of relacorilant and other product candidates, including their clinical attributes, regulatory approvals, mandates, oversight and other requirements; and the scope and protective power of our intellectual property. These and other risks are set forth in our SEC filings, which are available at our website and the SEC’s website.
In this press release, forward-looking statements include those concerning the development of relacorilant as a treatment for ovarian cancer, including its clinical attributes, regulatory approvals, mandates and oversight, and other requirements; the potential for relacorilant plus nab-paclitaxel to become a standard of care for patients with recurrent platinum-resistant ovarian cancer; our planned meetings with the Food and Drug Administration (FDA); our planned Phase 3 trial, including its design and start date; and the scope and protective power of our intellectual property. We disclaim any intention or duty to update forward-looking statements made in this press release.
CONTACT:
Corcept Therapeutics
Investor Relations
ir@corcept.com
www.corcept.comGraphics accompanying this announcement is available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/0c6c3a39-1cd5-4ace-83fe-82f15ff7d3a7https://www.globenewswire.com/NewsRoom/AttachmentNg/f5eefc78-3c7c-4d60-8acb-448ea42695d4
1 American Cancer Society (www.cancer.org)
2 Clarivate | Decision Resources Group Ovarian Cancer Market Forecast Dashboard - December 2021 (www.clarivate.com)
3 Therapeutic Advances in Medical Oncology (Luvero et al. 2014)
4 Journal of Clinical Oncology (Pujade-Lauraine et al. 2014)
5 Preliminary results from this trial were presented at the European Society for Medical Oncology (ESMO) Congress 2021. The ESMO presentation is available at www.corcept.com/research-pipeline/publications. Additional information about the study (NCT03776812) can be obtained at www.ClinicalTrials.gov.
- Addition of relacorilant to nab-paclitaxel reduces risk of death by 33 percent compared to treatment with nab-paclitaxel alone (p-value: 0.066), without increased side effects